Trending...
- Detroit Hip-hop Takes Center Stage: Historic Official Sxsw Showcase Spotlights The City's Rising Music Economy - 127
- Training Lofts Launches $1,099 Unlimited Training Membership Featuring Semi-Private Coaching, Nutrition Support, and Recovery Services - 119
- Pastor Saeed Abedini Releases THE TRUTH – Volume 1, A Deeply Personal Story of Faith, Struggle, and Redemption - 112
FAYETTEVILLE, Ark. - Michimich -- Lineus Medical is now officially registered in the United Kingdom, enabling the company to begin distributing SafeBreak® Vascular within the UK healthcare market. UK registration represents an important step in Lineus Medical's international expansion strategy, further extending access to SafeBreak Vascular for hospitals and clinicians on a global scale. With regulatory requirements in place, Lineus Medical can now work with distribution partners to bring its breakaway IV technology to the UK.
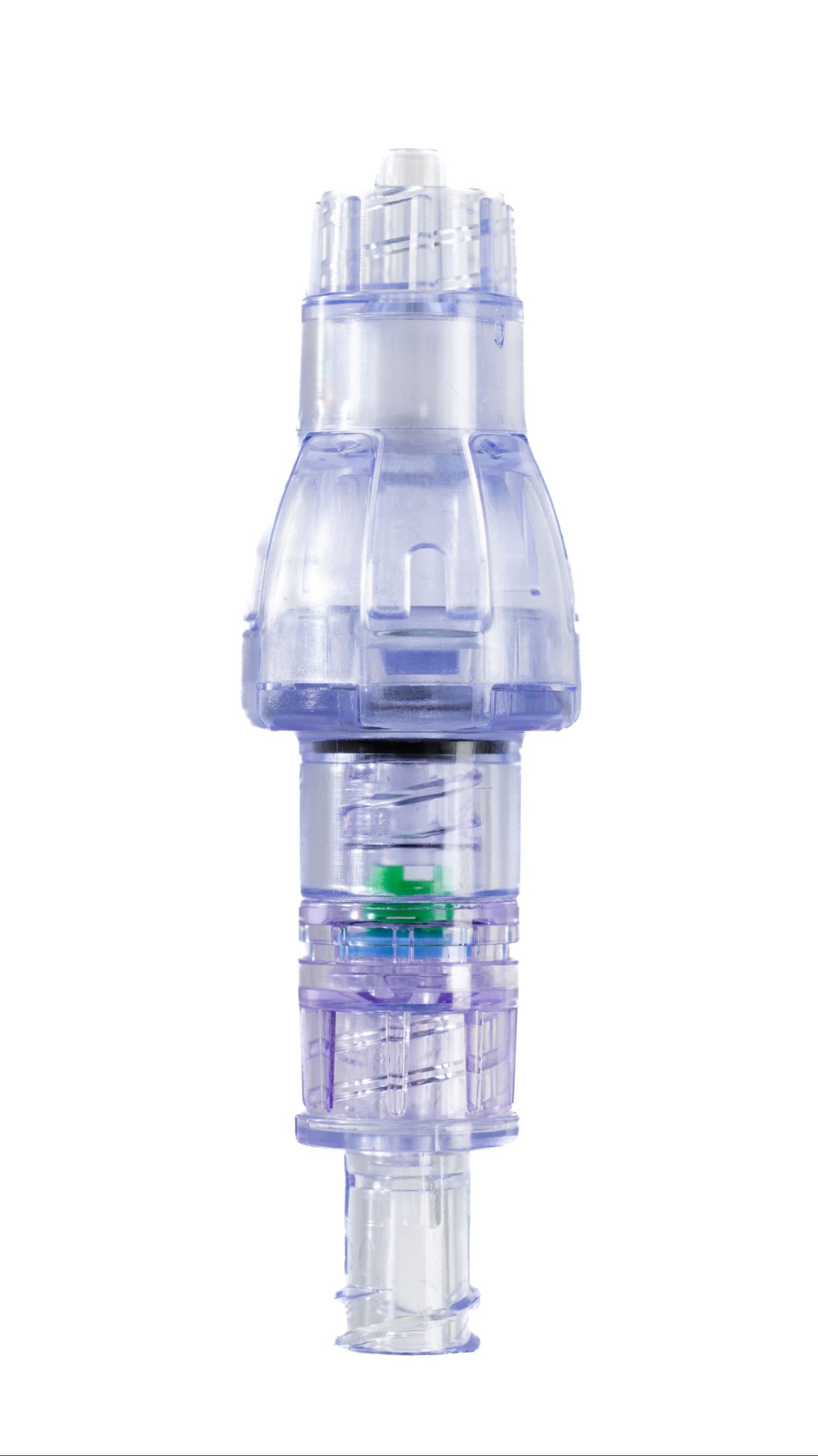
SafeBreak Vascular is a breakaway device for IV lines clinically proven to reduce IV complications.¹ When a harmful force is placed on the line, SafeBreak intentionally separates to remove the damaging force and protect the patient's IV. When separation occurs, valves on both ends of the device close to prevent medication spills from the pump and blood loss from the patient. Patients avoid additional needlesticks, nurses save time, and hospitals save money.¹
More on Michimich.com
"Completing UK registration is another meaningful milestone as we continue to expand access to SafeBreak globally," said Vance Clement, CEO of Lineus Medical. "Each new market brings us closer to our mission of removing the pains associated with IV lines and improving patient safety across healthcare systems worldwide. UK registration allows SafeBreak to reach another 69 million people."
"This registration allows us to move forward with distribution planning in the UK and supports our broader international commercialization efforts," said Larry Hayes, Chief Commercial Officer of Lineus Medical. "We are focused on working with the right partners to ensure SafeBreak Vascular is accessible to clinicians who are looking to reduce IV complications and improve care at the bedside."
About Lineus Medical:
Lineus Medical is the developer of SafeBreak® Vascular, a breakaway technology proven to reduce IV restarts and IV complications.1 Our mission is to remove the pains associated with medical lines. More information about Lineus Medical can be found at www.lineusmed.com. Follow Lineus Medical on LinkedIn, Facebook, and Instagram.
References
SafeBreak Vascular is a breakaway device for IV lines clinically proven to reduce IV complications.¹ When a harmful force is placed on the line, SafeBreak intentionally separates to remove the damaging force and protect the patient's IV. When separation occurs, valves on both ends of the device close to prevent medication spills from the pump and blood loss from the patient. Patients avoid additional needlesticks, nurses save time, and hospitals save money.¹
More on Michimich.com
- AAACF Celebrates Repaid Impact Investments, Renewing Capital for Washtenaw County Communities
- Roofman USA Expands Michigan Coverage, Ensuring Homes Across the State Stay Protected
- Roofman USA Highlights Key Features of Energy-Efficient Roofing for Ann Arbor Homeowners
- SMAA Highlights Unified Movement Principles in Jujutsu and Judo Through Educational Scholarship
- Compressed Gas Technologies, INC. Explains Key Differences Between Membrane and PSA N2 Generation
"Completing UK registration is another meaningful milestone as we continue to expand access to SafeBreak globally," said Vance Clement, CEO of Lineus Medical. "Each new market brings us closer to our mission of removing the pains associated with IV lines and improving patient safety across healthcare systems worldwide. UK registration allows SafeBreak to reach another 69 million people."
"This registration allows us to move forward with distribution planning in the UK and supports our broader international commercialization efforts," said Larry Hayes, Chief Commercial Officer of Lineus Medical. "We are focused on working with the right partners to ensure SafeBreak Vascular is accessible to clinicians who are looking to reduce IV complications and improve care at the bedside."
About Lineus Medical:
Lineus Medical is the developer of SafeBreak® Vascular, a breakaway technology proven to reduce IV restarts and IV complications.1 Our mission is to remove the pains associated with medical lines. More information about Lineus Medical can be found at www.lineusmed.com. Follow Lineus Medical on LinkedIn, Facebook, and Instagram.
References
- Data on file.
Source: Lineus Medical
Filed Under: Manufacturing
0 Comments
Latest on Michimich.com
- American Plastic Solutions Announces Versatile Plastic Plating Tanks and Enclosures
- Midwest Enviro Solutions Urges Homeowners & Businesses to Recognize Symptoms of Poor Indoor Air Qua
- $317M Revenue and a Clear Path to $1B: $IQST is Positioned for a Major Profitability Inflection
- Michigan Computer Supplies Shares Simple Maintenance Tips to Extend Office Printer Life & Reduce Em
- ASI Hosts 2026 Executive Business Summit for Global Partner Community
- Pastor Saeed Abedini Releases THE TRUTH – Volume 1, A Deeply Personal Story of Faith, Struggle, and Redemption
- New Book Warring From the Standpoint of the Throne Room Calls Believers to Pray From Victory
- Scotch Whisky Market Dislocation Creates Compelling Entry Opportunity for Long-Term Investors
- Peccioli Becomes New Orleans: In July 2026, the magic of jazz comes to Tuscany
- Michigan Debt Collection Agency Helps Businesses Create Airtight Credit Policies
- Michigan Collection Services Firm Describes Different Types of Defendants
- Howell-Based Professional Lawn Care Starts Crabgrass Prevention!
- Local Saline DJ Expands Services to Greater Grand Rapids Area
- $6 Million Funding Secured as Retail Expansion, Operational Streamlining, and Asset-Light Strategy Position the Company for Accelerated Growth $SOWG
- The "Unsexy" Business Quietly Creating 130+ New Entrepreneurs Across America — From Alaska to Puerto Rico
- Veteran Launches GTG Energy: Nicotine-Free Pouch as Americans Rethink Addiction, Focus, and What Fuels Performance
- RecallSentry™ App Launch — Your Home Safety Hub — Free on iOS & Android
- Award-Winning Director Crystal J. Huang's Under-$50K Film "The Ritual House" Wins Best Horror Feature at Golden State Film Festival
- Grads aren't getting hired — here's what we're doing about it
- K2 Integrity Enhances Technology Capabilities Through Acquisition of Leviathan Security Group